Description
This Bovine Foot-and-Mouth Disease Virus-Like Particle (VLP) Pentavalent Vaccine is a next-generation livestock vaccine designed to provide broad-spectrum protection against five major FMD serotypes affecting cattle and sheep globally.
The vaccine includes:
- Type O (PanAsia2 lineage)
- Type A (Iran05 lineage)
- Type Asia 1 (Sindh-8 lineage)
- Type SAT1
- Type SAT2
Unlike traditional inactivated vaccines, this product utilizes virus-like particle (VLP) technology, which offers:
- Higher safety (no live virus involved)
- Strong immunogenicity
- Improved antigen stability
This makes it particularly suitable for:
- High-risk FMD regions
- Cross-border livestock trade zones
- Large-scale cattle and sheep farming operations
With increasing genetic diversity of FMD viruses worldwide, this multi-serotype formulation ensures maximum field coverage and risk reduction, helping farms maintain biosecurity and production stability.
Vaccination Program
- Initial vaccination for calves at ~90 days of age
- Booster after 30 days
- Routine revaccination every 4–6 months
- Suitable for:
- Endemic areas
- High-risk regions
- Intensive livestock systems
Dosage & Administration
- Route: Intramuscular injection
- Dosage:
- Cattle: 2.0 mL per head
- Sheep: 1.0 mL per head (half dose)
Composition
Each dose contains:
- Type O (O/Tur/5/2009 PanAsia2): 52.5 μg VLP
- Type A (A/Tur/06-20 Iran05): 52.5 μg VLP
- Type Asia 1 (Asia1/Pak/08 Sindh-8): 52.5 μg VLP
- Type SAT1: 52.5 μg VLP
- Type SAT2 (Eritrea 98): 52.5 μg VLP
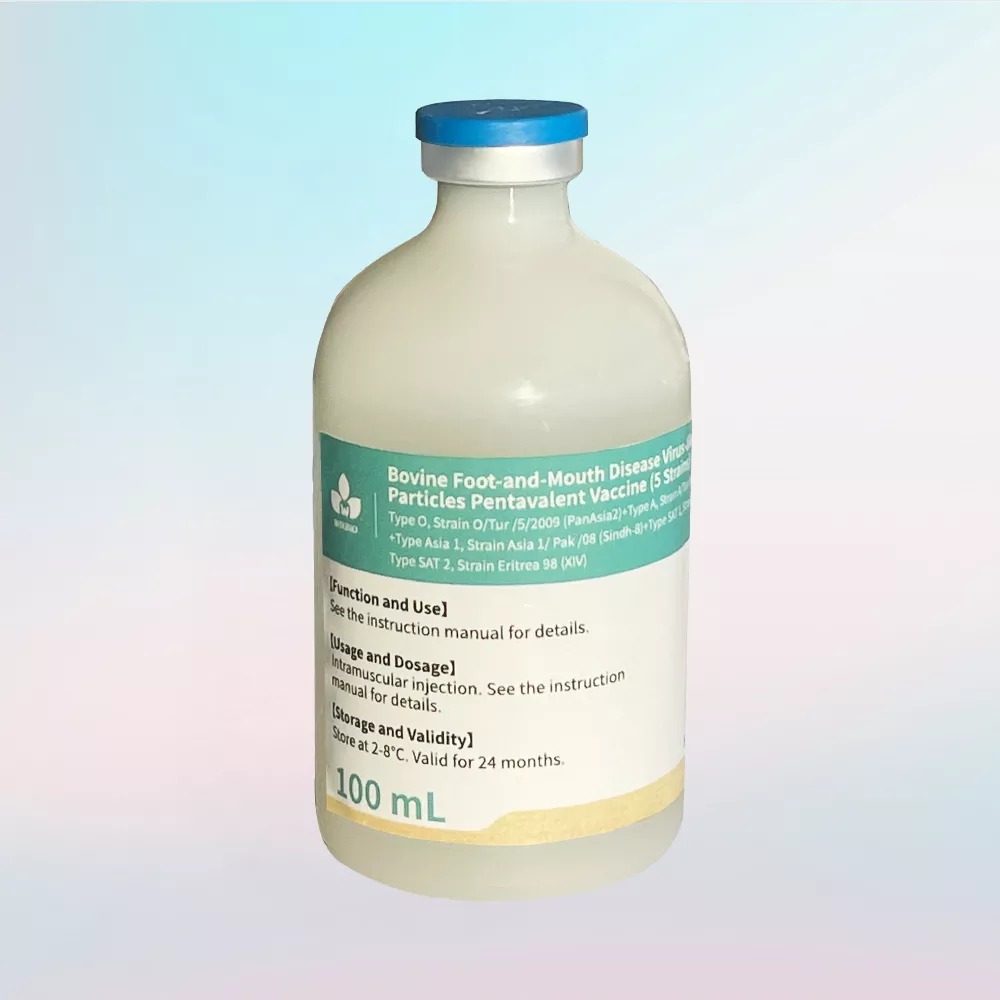
Product Characteristics
- Milky white oil emulsion
- VLP-based high safety profile
- Broad serotype coverage
- Strong and stable immune response
Safety & Precautions
- Use only in healthy livestock
- Avoid vaccination in:
- Early/late pregnancy
- Recently transported animals
- Do not freeze; maintain cold chain (2–8°C)
- Shake well before use
- Use within the same day after opening
- Follow strict aseptic injection procedures
- Monitor animals post-vaccination for allergic reactions
Packaging & Specification
- Specification:
- 100 mL/bottle
- Packaging:
- 100 bottles/carton
Storage & Shelf Life
- Storage: 2–8°C
- Shelf Life: 24 months