Product Description
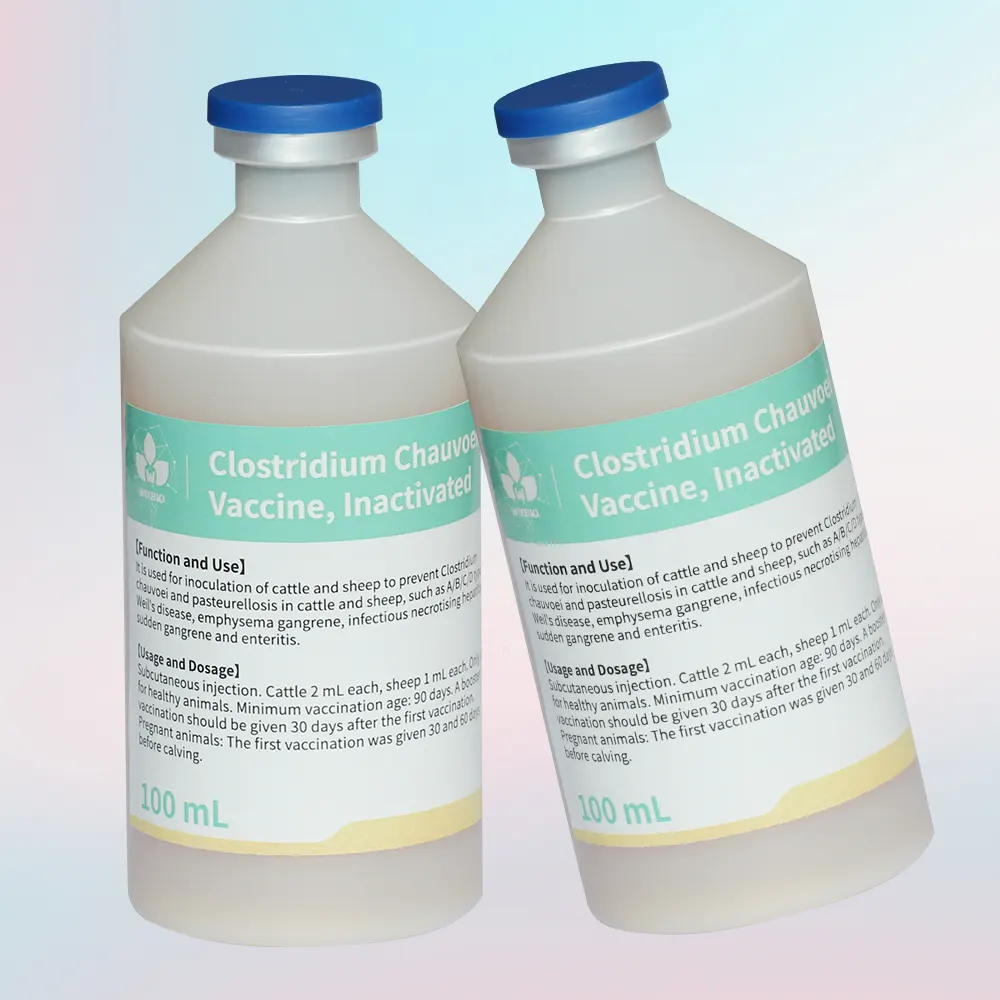
This inactivated multivalent vaccine is designed to provide broad-spectrum protection against major clostridial infections and pasteurellosis in cattle and sheep, which are among the most economically significant diseases in ruminant production systems.
Clostridial diseases are typically acute, highly fatal, and often associated with environmental exposure to spores. These include blackleg (caused by Clostridium chauvoei), enterotoxemia (Clostridium perfringens types A–D), malignant edema (Clostridium septicum), infectious necrotic hepatitis (Clostridium novyi), and other toxin-mediated conditions. Additionally, pasteurellosis caused by Pasteurella multocida leads to respiratory disease and septicemia, particularly under stress conditions.
The inclusion of tetanus toxoid further enhances protection by preventing toxin-mediated neuromuscular disease, especially in animals undergoing procedures such as castration or lambing.
With a comprehensive antigen profile, this vaccine is well suited for integrated herd health management programs, reducing mortality, improving productivity, and minimizing economic losses.
Protective immunity develops after completion of the primary vaccination course and lasts for up to 12 months, supporting annual vaccination strategies.
Immunization Program
- Primary Vaccination:
- First dose at ≥ 90 days of age
- Booster dose after 30 days
- Pregnant Animals:
- Two doses: 60 days and 30 days before parturition
- Revaccination:
- Annual booster
Dosage & Administration
- Route: Subcutaneous injection
- Cattle: 2.0 mL per animal
- Sheep: 1.0 mL per animal
Antigen Composition
Contains inactivated or toxoid components of:
- Clostridium chauvoei
- Clostridium septicum
- Clostridium perfringens (Types A, B, C, D)
- Clostridium sordellii
- Clostridium novyi type B
- Pasteurella multocida (Group A & B)
- Tetanus toxoid
Product Characteristics
- Appearance: Milky white uniform emulsion
- Stable multivalent formulation
- Designed for broad-spectrum protection
Safety & Adverse Reactions
- Generally well tolerated in healthy animals
Possible reactions:
- Rare hypersensitivity reactions (shock)
- Immediate treatment with epinephrine recommended
- Do not use in:
- Animals with fever
- Animals under stress
Packaging & Specification
- Specification: 100 mL/bottle
- Packaging: 100 bottles/carton
Storage & Shelf Life
- Storage: 2–8°C, protected from light
- Do NOT freeze
- Shelf Life: 24 months
Precautions
- Use only in healthy animals
- Avoid vaccination during stress conditions
- Maintain aseptic injection procedures
- Follow proper herd vaccination protocols