Advanced FMD Subunit Vaccine Description
This quadrivalent vaccine is developed using advanced virus-like particle (VLP) technology, representing the next generation of foot-and-mouth disease (FMD) vaccines.
Unlike traditional inactivated vaccines, this product contains non-infectious VLP antigens, which structurally mimic the native virus but contain no viral genetic material. This ensures excellent safety with zero risk of viral replication or reversion, while maintaining strong immunogenicity.
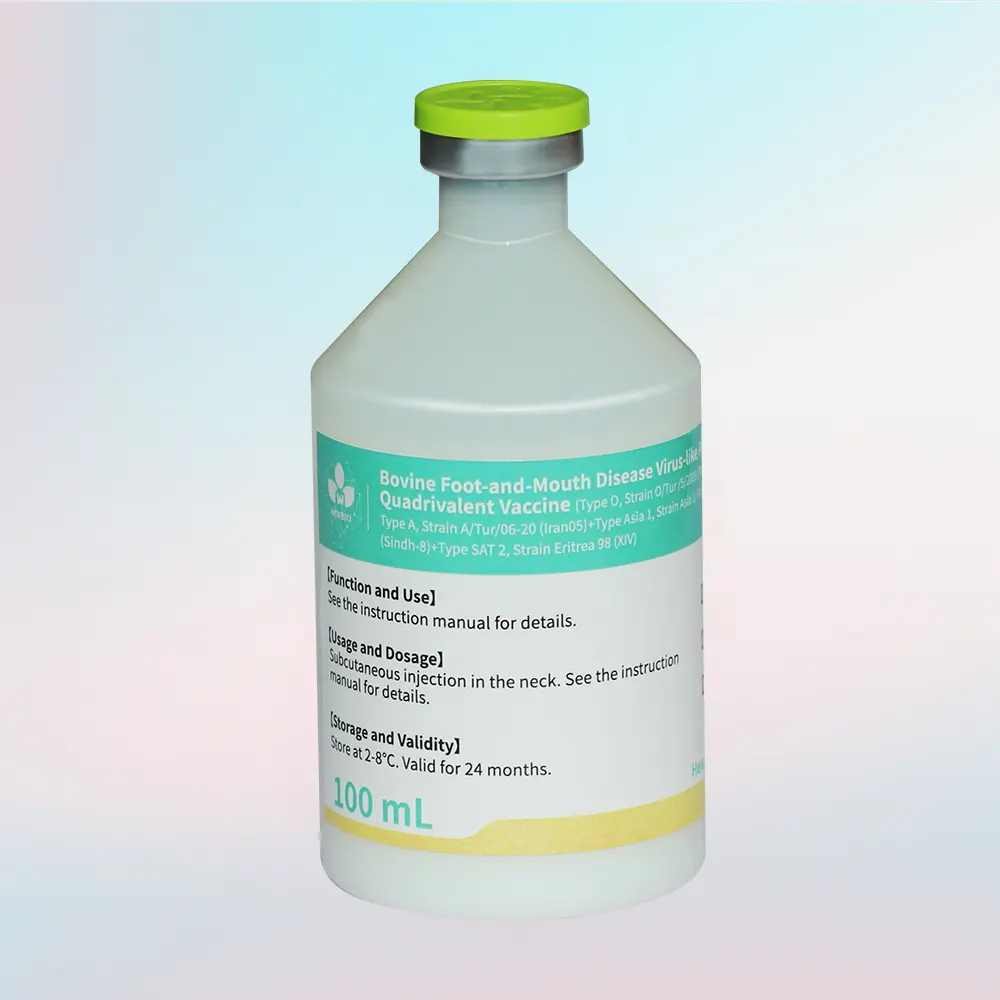
The vaccine provides protection against four major FMD serotypes of global importance:
- Type O (PanAsia2 lineage)
- Type A (Iran05 lineage)
- Type Asia 1
- Type SAT 2
These serotypes are responsible for most transboundary FMD outbreaks across Asia, the Middle East, and Africa. The inclusion of SAT 2 significantly enhances protection for regions with emerging or complex epidemiological risks.
Immunity develops within 3 weeks after vaccination and lasts for up to 6 months, making this vaccine suitable for both routine immunization and strategic disease control programs, including high-biosecurity farms and international livestock trade systems.
Immunization Program
- Calves:
- First dose: at approximately 90 days of age (2.0 mL)
- Booster: 30 days later
- Routine Revaccination:
- Every 4–6 months
- Sheep:
- Half dose of cattle regimen
Dosage & Administration
- Route: Intramuscular injection
- Cattle: 2.0 mL per dose
- Sheep: 1.0 mL per dose
Antigen Composition (VLP Content)
Each dose contains:
- Type O (O/Tur/5/2009, PanAsia2): 52.5 μg VLP protein
- Type A (A/Tur/06-20, Iran05): 52.5 μg VLP protein
- Type Asia 1 (Asia 1/Pak/08): 52.5 μg VLP protein
- Type SAT 2 (Eritrea 98, XIV): 52.5 μg VLP protein
Product Characteristics
- Appearance: Milky-white emulsion
- Non-infectious VLP formulation
- High antigen purity and stability
Safety & Adverse Reactions
General safety profile:
- Typically no significant adverse reactions
Possible mild reactions:
- Injection site swelling
- Temporary fever
- Reduced feed intake (1–2 days)
These effects usually resolve within 3–5 days.
Rare severe reactions:
- Hypersensitivity:
- Restlessness
- Rapid breathing
- Muscle tremors
- Mucosal congestion
- In rare cases:
- Death if untreated
- Possible abortion in pregnant animals
Emergency management:
- Immediate administration of epinephrine
- Supportive veterinary care
Packaging & Specification
- Specification: 100 mL/bottle
- Packaging: 100 bottles/carton
Storage & Shelf Life
- Storage: 2–8°C
- Protect from light
- Do NOT freeze
- Shelf Life: 24 months
Precautions
- Use only in healthy livestock
- Avoid vaccination in:
- Early and late pregnancy
- Pre-weaning animals
- Animals under transport stress
- Bring vaccine to room temperature and shake well before use
- Use opened vaccine within the same day
- Follow strict aseptic procedures:
- Disinfect injection sites
- Use sterile equipment
- Replace needles between animals
- Vaccination should be performed by trained professionals
- In outbreak control:
- Vaccinate in order: safe area → threatened area → epidemic area
- Movement restriction:
- Allow livestock movement only after 21 days post-vaccination in non-epidemic areas
- Combine vaccination with:
- Biosecurity
- Quarantine
- Disinfection protocols