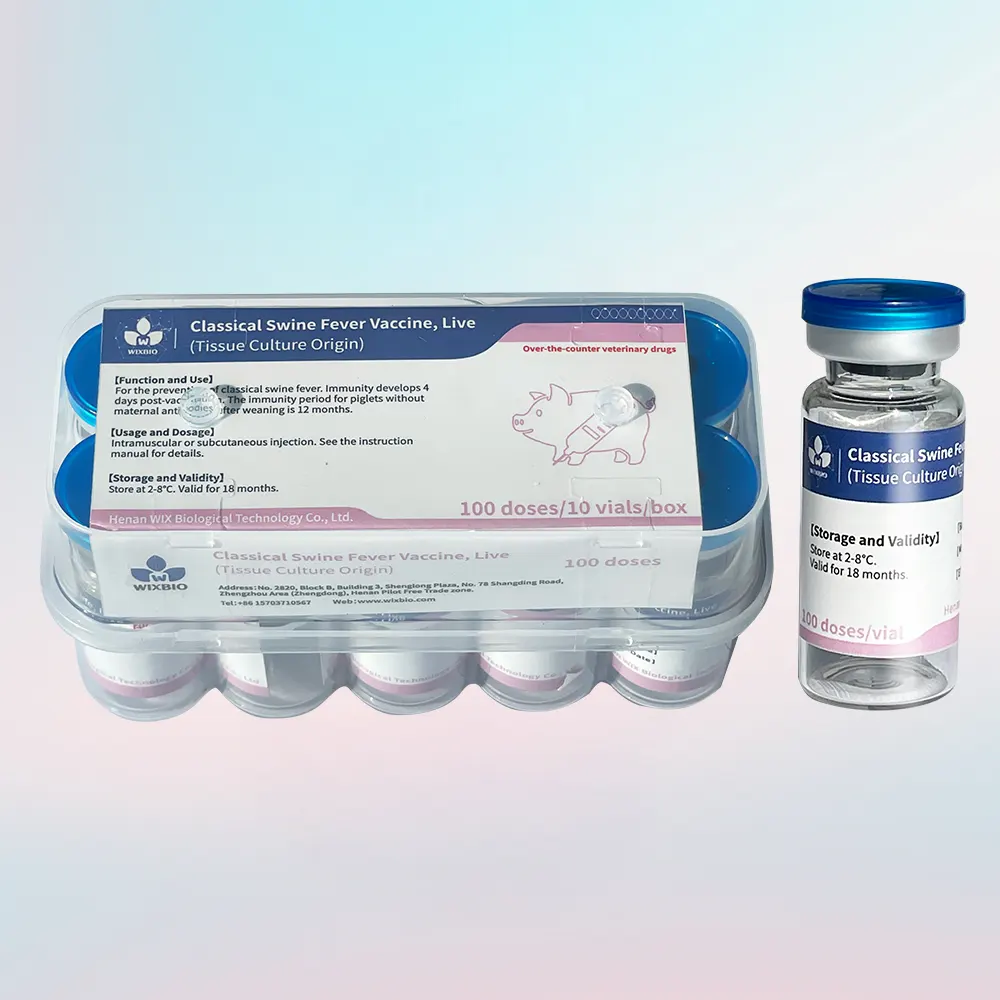
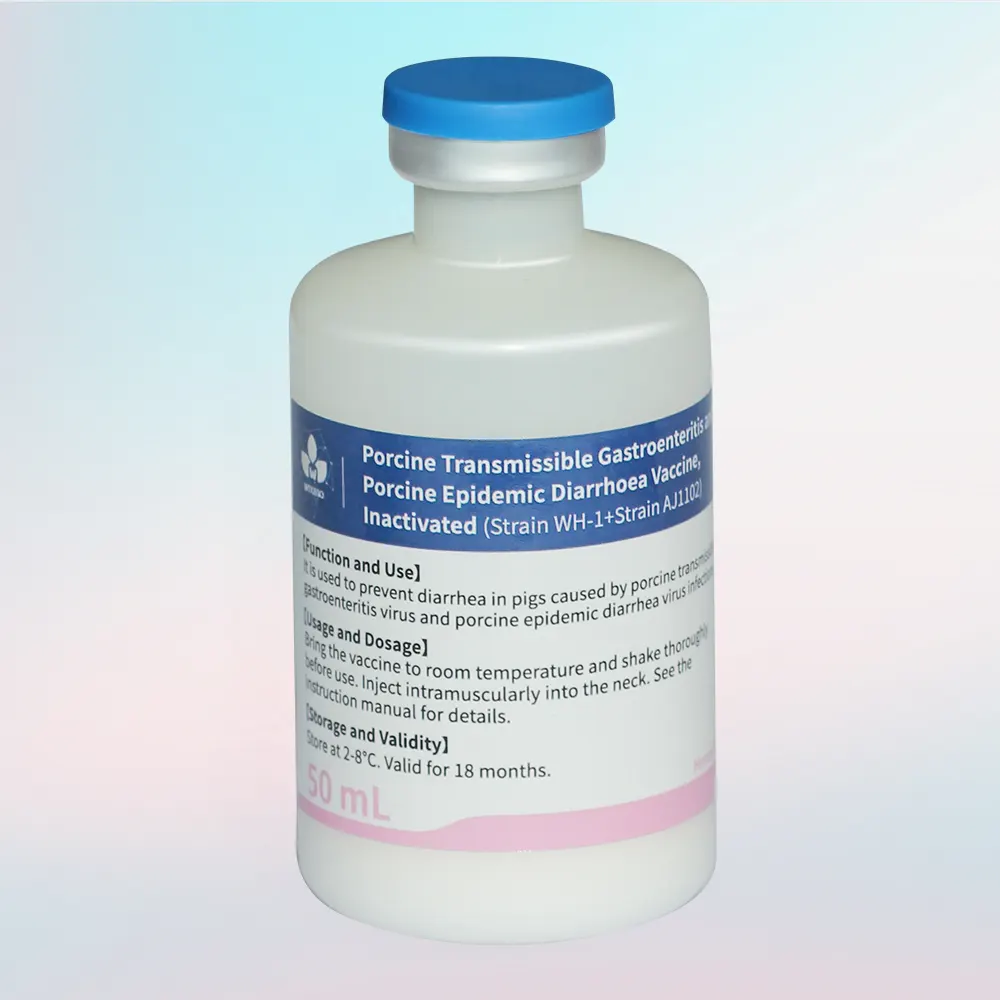
Inactivated FMD Vaccine Overview
This vaccine is a bivalent inactivated Foot-and-Mouth Disease (FMD) vaccine specifically designed for pigs, targeting Type O strains prevalent in Asia, including:
- O/Mya98/XJ/2010 (Southeast Asia topotype)
- O/GX/09-7 (variant strain)
Key Technical Features:
- High antigen payload: ≥600 ng per strain per dose
- Potency: ≥6 PD50 per dose (for each strain)
- Virus content before inactivation: ≥10⁷.0 TCID50/mL
Benefits:
- Strong cross-protection against circulating Type O strains
- Suitable for endemic and high-risk regions
- Designed for intensive pig farming systems
Immunization Program
- Suitable for growing pigs and adult pigs
- Can be used in routine vaccination programs and regional disease control strategies
Dosage & Administration
Route: Intramuscular injection
- Injection Site: Behind the ear root
| Pig Weight | Dosage |
|---|---|
| 10–25 kg | 1 mL (½ dose) |
| >25 kg | 2 mL (1 dose) |
Administration Notes:
- Multi-point injection recommended for piglets
- Use one needle per animal to prevent cross-contamination
Composition
- Inactivated FMD virus Type O (Strain O/Mya98/XJ/2010)
- Inactivated FMD virus Type O (Strain O/GX/09-7)
- Each strain ≥600 ng antigen per dose
- Potency ≥6 PD50 per strain
Product Characteristics
- Light pink or milky white viscous emulsion
- Stable oil-emulsion formulation for prolonged immunity
Safety & Adverse Reactions
General Reactions:
- Mild swelling at injection site
- Temporary fever
- Reduced appetite for 1–2 days
Severe (Rare) Reactions:
- Hypersensitivity (respiratory distress, tremors, nasal bleeding)
- Possible abortion in pregnant sows
- Immediate treatment with epinephrine or dexamethasone required
Precautions
- Store and transport at 2–8°C, do not freeze
- Avoid vaccination in:
- Sick or weak pigs
- Stressed animals (transport, environmental changes)
- Late pregnancy (use with caution)
- Shake well and restore to room temperature before use
- Use aseptic injection techniques (one needle per pig)
- Vaccination should be performed by trained professionals
- Follow vaccination order:
- Safe areas → Threatened areas → Endemic areas
- Movement of animals allowed 21 days after vaccination
- Maintain strict biosecurity measures (disinfection, quarantine)
- Keep detailed vaccination records
- Dispose of vaccine waste according to biosafety regulations
Packaging & Specification
- Specification: 100 mL/bottle
- Packaging: 100 bottles/carton
Storage & Shelf Life
- Storage: 2–8°C, protect from light
- Shelf Life: 24 months