Product Name
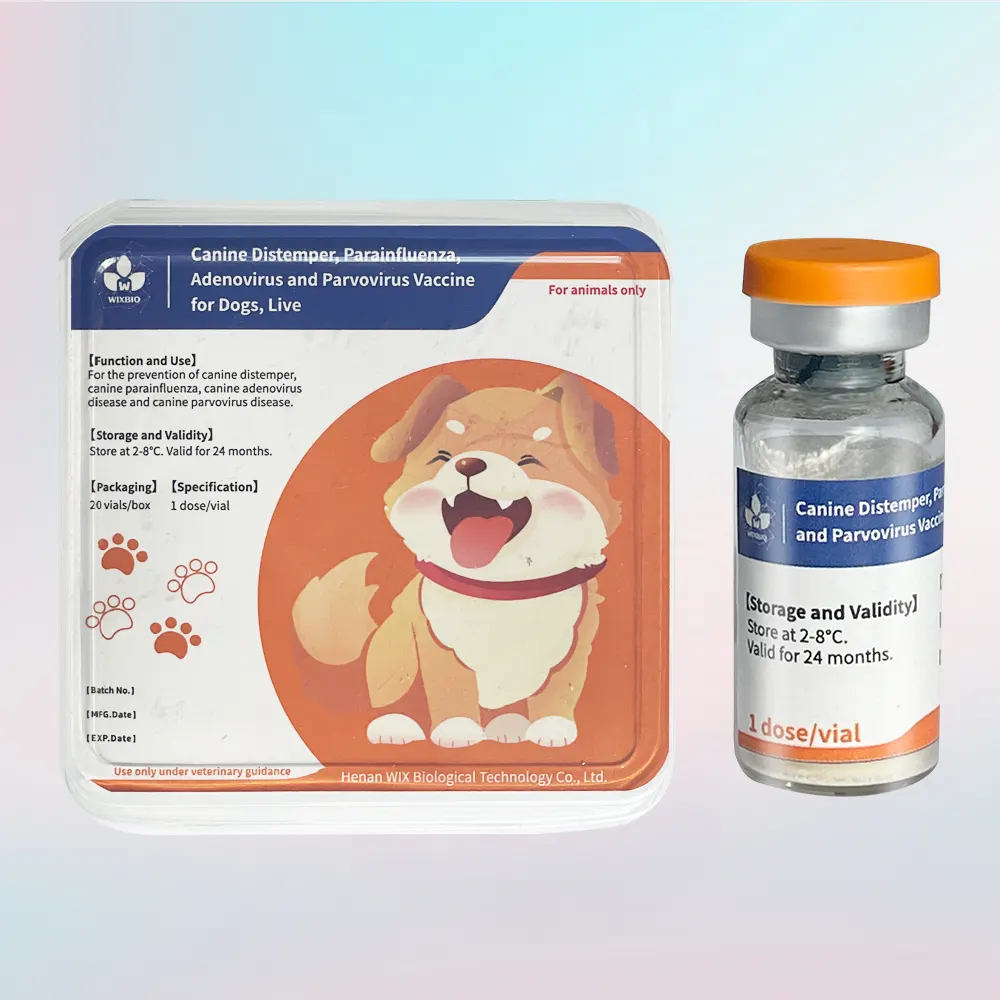
Canine Distemper, Parainfluenza, Hepatitis and Parvovirus Vaccine, Live
Main Composition and Content
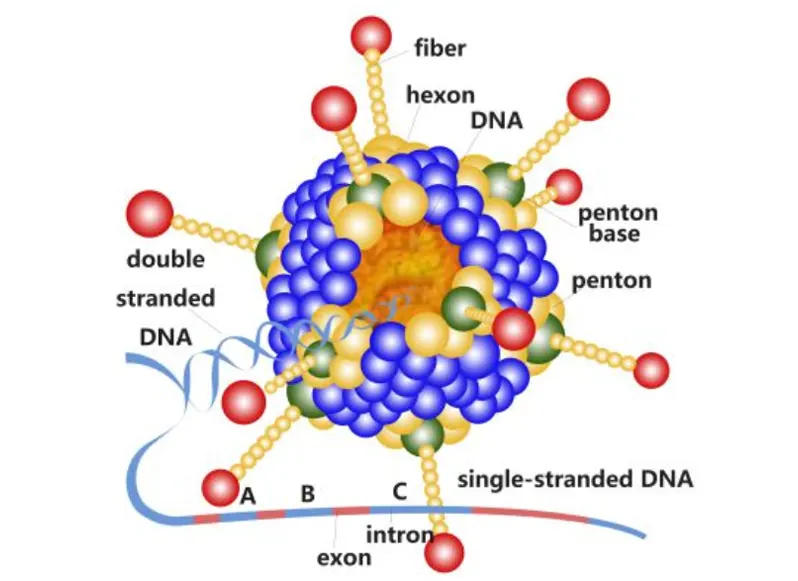
Each dose contains at least 104.0 TCID50 of attenuated canine distemper virus(strain CDV/R-20/8), attenuated canine parainfluenza virus(strain CPIV/A-20/8), attenuated canine adenovirus(strain YCA18), and attenuated canine parvovirus(strain CR86106).
Properties
Slightly yellowish-white, spongy, loose clumps that easily detach from the vial wall. Upon addition of diluent, it quickly dissolves into a clear, pink liquid.
Function and Use
For the prevention of canine distemper, canine parainfluenza, canine adenovirus disease and canine parvovirus disease. The duration of immunity is 12 months.
Usage and Dosage
Intramuscular injection. Dilute with water for injection to 2.0 mL per dose. For weaned puppies, administer three consecutive times of 2.0 mL each at 21-day intervals. For adult dogs, administer twice a year, with an interval of 21 days, 2.0 mL each time.
Precautions
- This vaccine is for preventive immunization of non-food dogs only. It must not be used for emergency vaccination or treatment during an active outbreak. Do not use in pregnant dogs.
- Dogs that have received immune serum should wait 7-14 days before receiving this vaccine.
- Useaseptic syringes for vaccination.
- Use the vaccine immediately after dilution.
- During the vaccination period, it is necessary to avoid mobilization, transportation, and sudden changes in breeding management conditions, and to prohibit contact with sick dogs.
- If an allergic reaction occurs after vaccination, immediately intramuscularly inject 0.5-1.0 mLof epinephrine hydrochloride injection.
- Treat the used vaccine vials, equipment, and remainedvaccines harmlessly.
Size
1 dose/vial
Packing
50 vials/box
Storage and Validity
Store at 2~8℃, and valid for 24 months.