Newcastle Disease, AI (H9), IBD & Fowl Adenovirus Vaccine Description
This Newcastle Disease, Avian Influenza (H9), Infectious Bursal Disease (IBD), and Fowl Adenovirus (FAdV) Inactivated Vaccine is a high-level multivalent poultry vaccine designed to provide comprehensive protection against four major disease groups affecting modern poultry production.
The vaccine integrates:
- Newcastle disease virus (rGM-VII strain)
- Avian influenza H9 subtype (WD strain)
- Infectious bursal disease antigen (rVP2 protein)
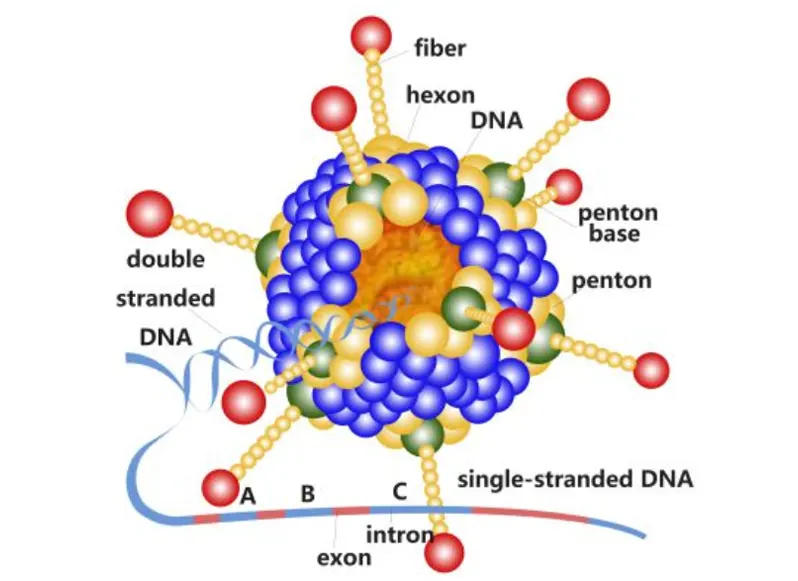
- Fowl adenovirus serotypes (FAdV-4, FAdV-8b, FAdV-11)
This broad-spectrum formulation targets both:
- Respiratory and systemic viral infections (ND, AI)
- Immunosuppressive diseases (IBD)
- Emerging adenovirus-associated diseases (hydropericardium syndrome, inclusion body hepatitis)
With increasing prevalence of FAdV outbreaks globally, this vaccine offers a strategic advantage by incorporating multiple adenovirus serotypes, ensuring wider field protection.
The oil-emulsion formulation provides strong and sustained immunity, while the combination design significantly reduces vaccination frequency, labor costs, and handling stress, making it ideal for intensive commercial poultry operations.
Vaccination Program
- Suitable for chickens ≥ 1 week old
- Can be integrated into intensive vaccination schedules
- Recommended for farms with:
- High disease pressure
- FAdV outbreak history
- Complex multi-pathogen risks
Dosage & Administration
- Route:
- Subcutaneous (neck)
- Intramuscular injection
- Dosage:
- 1–4 weeks: 0.3 mL per bird
- 4 weeks: 0.5 mL per bird
Composition
Each 0.5 mL dose contains:
- NDV (rGM-VII): ≥ 10⁸.² EID50
- H9 (WD strain): ≥ 10⁸.² EID50
- IBD (rVP2): AGP titer ≥ 1:48
- FAdV-4: ≥ 10⁷.² TCID50
- FAdV-8b: ≥ 10⁷.² TCID50
- FAdV-11: ≥ 10⁷.² TCID50
Product Characteristics
- Milky white uniform oil emulsion
- High antigen content
- Broad serotype coverage (especially FAdV)
- Designed for high-risk environments
Safety & Precautions
- Use only on healthy chickens
- Do not freeze
- Do not use if emulsion is broken
- Shake well before use
- Use within the same day after opening
- Maintain aseptic injection procedures
Packaging & Specification
- Specifications:
- 250 mL/bottle
- 500 mL/bottle
- Packaging:
- 40 bottles/carton
- 20 bottles/carton
Storage & Shelf Life
- Storage: 2–8°C
- Shelf Life: 24 months